How to clean ORP and pH probes
Let’s face it: eventually the probes in a chemical controller will get dirty. Maybe it’s some calcification, or the usual oily organics that stick to the bulb. Whatever has dirtied the probe, you can bet your water chemistry readings will be distorted. In this article, we cover an easy way to clean ORP and pH probes.
Clean probes are essential to controllers
Pool chemical controllers rely on probes to tell the system what your water chemistry is. There are probes for Oxidation-Reduction Potential (ORP), Total Dissolved Solids (TDS), Free Available Chlorine (FC or FAC), Total Available Chlorine (TAC), and of course, the most common type, pH probes. As far as we know, every controller has at least a minimum of a pH probe, but most controllers also have ORP. The fancier controllers like the Prominent DCM5 have free chlorine and total chlorine too, which allow the controller to calculate combined chlorine (CC) in real time.


When probes get dirty, their accuracy is diminished. Since these probes are submerged in flowing water, contaminants in the water itself can adhere to the probes themselves, and that’s the purpose of this article. Just imagine if your probes were sitting in a sample that looked like this:
How accurate can those probes really be? Just look at the sample water! As a pool operator, this should be unacceptable, because the controller is not going to have accurate inputs, which means chemistry is inevitably going to stray from the desired set points.
Cleaning procedure
You will need:
- Three disposable or washable cups, or plastic containers, bowls, etc.
- Distilled water (bottled water is fine)
- A tiny amount of muriatic acid (use an eye dropper or syringe)
- Safety gloves/glasses
- SimplyPURE enzymes
- Toothbrush (soft bristles)
First, fill two cups about halfway with distilled water. Add a few drops of Muriatic Acid to one of them using an eye dropper. The second cup should remain clean. In the third cup, add about 2 fl.oz. of SimplyPURE enzyme.
Second, close off the water flow to the probes, and follow the controller manufacturer’s recommended procedure before removing the probes. Most controller setups have an easy-to-find valve that simply shuts off the water to the probes.
Then, remove the probes that need to be cleaned, following the controller manufacturer’s recommended procedure.
Dip the probe in the enzyme, then brush with the toothbrush gently to remove oils and other ‘gunk’. This step alone may clean the probe sufficiently, but if there is calcium buildup remaining, proceed to the next step. If not, rinse the probe in the distilled water, and put it back where it belongs.

If calcification is on the probe, submerge the probe in the first cup that has a few drops of acid in it. Keep it in for about 30-40 seconds. Brush with the toothbrush to remove calcium buildup. Repeat the enzyme wash and acid wash as necessary until the buildup is gone. Then rinse in the distilled water before putting the probe back.
Prevention
Preventing calcium and organic buildup on probes can be as simple as using Natural Pool Products on a regular maintenance basis. NaturallyFREE is a scale inhibitor which will prevent calcium from forming on the probes (and should help prevent scale anywhere else in the system too). SimplyPURE enzymes will address the non-living organics, which should also help keep the probes clean, much like it helps keep tile lines clean.
Hopefully this procedure is easy enough to follow, and we hope to release a helpful how-to video on the procedure in the future. If you have more questions, please contact us, or your local NPP dealer.













 As mentioned before, without an enzyme, chlorine is held responsible for all of this bather waste. And while chlorine is excellent at disinfection, it gets used up quickly and inefficiently when fighting bather waste and oils. Enzymes, however, devour them. Enzymes are an affordable, safe and easy supplement to help keep your
As mentioned before, without an enzyme, chlorine is held responsible for all of this bather waste. And while chlorine is excellent at disinfection, it gets used up quickly and inefficiently when fighting bather waste and oils. Enzymes, however, devour them. Enzymes are an affordable, safe and easy supplement to help keep your  The initial dose of SimplyPURE enzymes is one quart per 10,000 gallons (32 fl.oz./10,000 gallons). This should be applied to either the surge tank or directly into the gutter. The idea is to put the enzymes directly into circulation so they spread around the pool quickly and evenly. Sure, you could pour them into the pool, but they will not circulate as fast.
The initial dose of SimplyPURE enzymes is one quart per 10,000 gallons (32 fl.oz./10,000 gallons). This should be applied to either the surge tank or directly into the gutter. The idea is to put the enzymes directly into circulation so they spread around the pool quickly and evenly. Sure, you could pour them into the pool, but they will not circulate as fast.

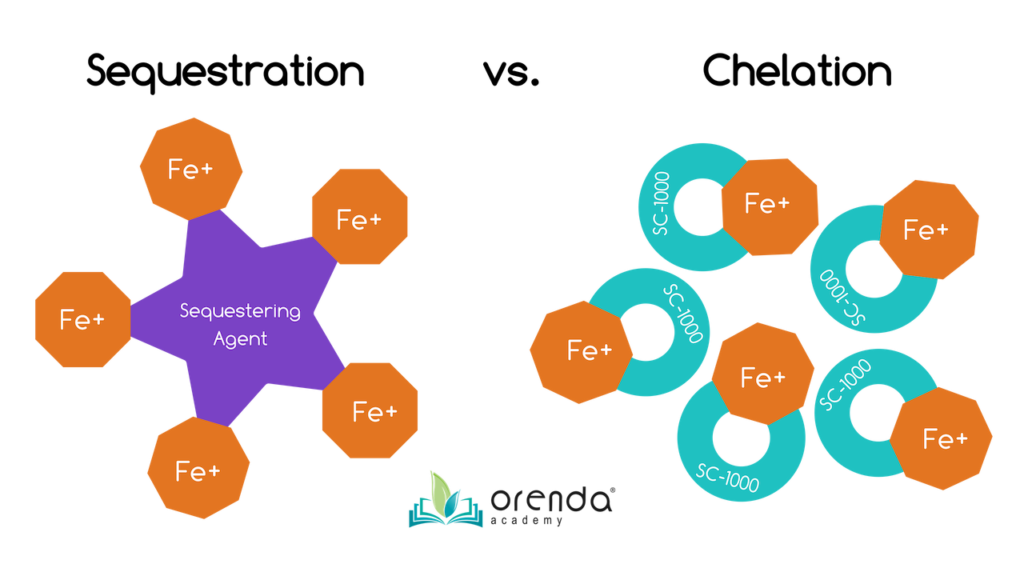
 Chelating Agent: NaturallyFREE
Chelating Agent: NaturallyFREE On the second day, once the pool is full, you will add the second chemical (usually bicarb), along with a purge dose of our enzyme,
On the second day, once the pool is full, you will add the second chemical (usually bicarb), along with a purge dose of our enzyme, 








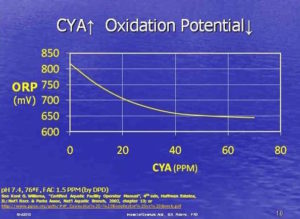
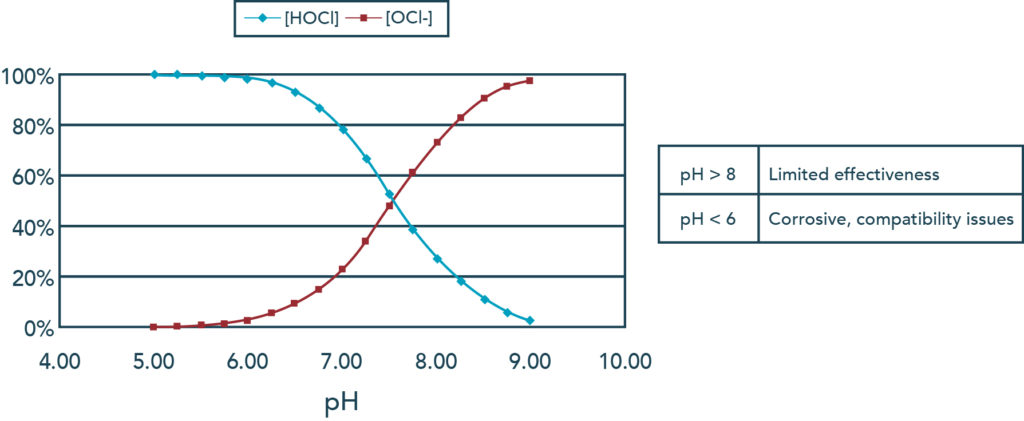
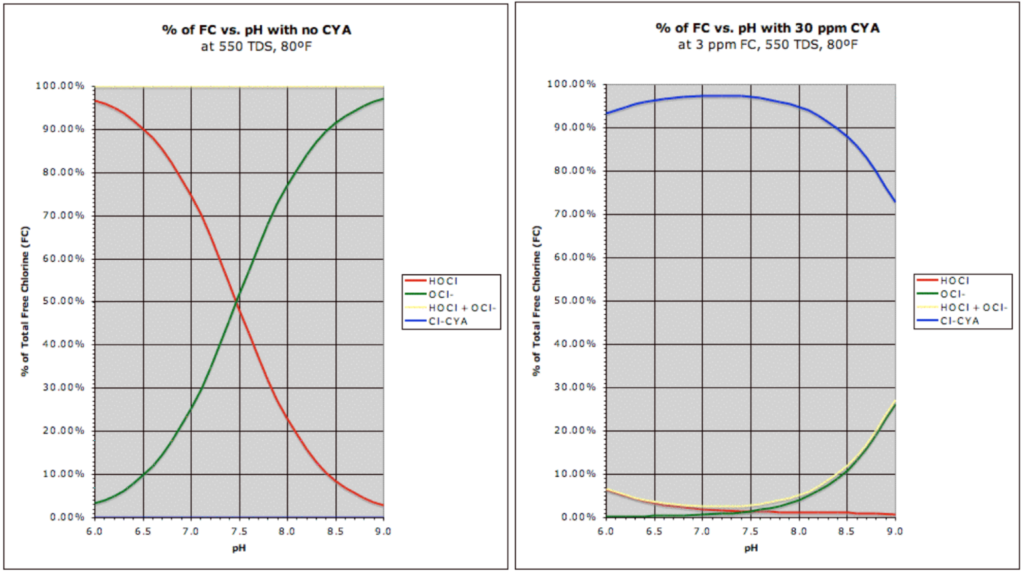
 Yes,
Yes, 


 But the question arose, “How can there be phosphates in the water? It’s literally filling up right now.” The answer is simple: the phosphates are in the tap water.
But the question arose, “How can there be phosphates in the water? It’s literally filling up right now.” The answer is simple: the phosphates are in the tap water.
