How to manage Iron in Swimming Pools
If you have ever had brown stains in your swimming pool, you are already all-too-familiar with iron problems. In this article, we discuss how iron gets into our water and why it creates stains. Then we will discuss options for managing iron to prevent problems like stains and excessive chlorine demand.
Metal Oxidation creates higher chlorine demand
Perhaps the main consequence of having iron and other metals in your water is increased chlorine demand. Metals are the easiest thing for chlorine to oxidize, and therefore the first things to be oxidized. As oxidants, metals like iron reduce chlorine rapidly. This is why at the beginning of the breakpoint chlorination curve (up to point A on the chart below), there is no noticeable increase of chlorine residuals until these “chlorine reducing compounds” like iron are conquered.

Pool operators with iron issues may notice a higher consumption of chlorine, but often it is overlooked. The cost may not be noticeably higher because the pool might be constantly introducing new water with iron in it. In other words, it’s the baseline, and nothing to compare it to. But rest assured, iron absolutely reduces free chlorine in water. Below we will discuss ways to control metals like iron, but first, let’s talk about where iron comes from.
How does iron get into water?
Iron is found in almost all natural water sources. According to a drinking water equipment manufacturer, LennTech, iron is in seawater, rivers, lakes and groundwater too. It up to those of us who manage and treat water to remove it. So iron usually gets into our swimming pools via the tap water. Certain areas of the United States have more iron in their tap water than others. For instance, the upper midwestern states (Ohio, Michigan, Indiana, Illinois, Wisconsin and Minnesota) are challenged with notorious iron problems. Swimming pools in those areas must deal with high levels of iron out of the faucet, or be plagued with iron staining.

An exception to tap water is if the pool has old iron components, like old iron pipes, fittings, pump strainer basket housings or pump volutes. If these components have been worn down (like having low pH water flowing through them long enough), iron is sure to find its way into the pool.
There are some exceptions for other metals too, like copper. Copper can get into water from products like copper algaecide, mineral disinfection systems, and from corroding heat exchangers or copper pipes. Pools have turned green from copper before, and sometimes it is mistaken for algae. But let’s get back to iron.
Dissolved iron in water is mainly present as ferrous hydroxide (Fe(OH)2+). We could try to explain the chemical formulas and reactions, but you can read these three sources if you are interested in that level of detail. Source 1, Source 2, or you can go down the Wikipedia rabbit hole as we did. But for this article here, we will try and keep this as simple as possible.

Usually, iron gets in our swimming pools in a dissolved, soluble state. This means it is mostly invisible and not yet oxidized. If drinking water is chlorinated or chloraminated, however, some of the iron may become oxidized and have a tinge of brown color to it. Iron, when oxidized, turns reddish-brown, and chlorine is consumed (reduced) in the process. If the iron was not yet oxidized in the pipes en route to the swimming pool, it sure will be when it is met with chlorine or a secondary oxidizer like ozone.
Oxidation is what creates staining.

Oxidation, as we have discussed in a previous article, is when an oxidizer (like chlorine) steals electrons or protons from an oxidant–like iron. So the key to preventing iron staining is to prevent iron from being oxidized. And to do that, we have a couple of options.
How to prevent iron staining
We can try to remove iron, either from the source water or from the pool itself, and/or we can chemically manage it by using a chelating agent or sequest.
Filter Iron out of the tap water
Iron filters exist and can be installed on pool fill lines. They do need to be replaced periodically; no filter has unlimited capacity. Such iron filters have a wide range in price depending on the removal rate you need, and the flow rate of your water. For instance, a 2″ fill line on a commercial pool will need a larger, more expensive iron filter than a garden hose filling a residential pool.
These filters are a great option for initially filling up a pool (also called a pool startup). The iron–and other metals that may be present in the fill water–will be filtered out prior to adding chlorine to the pool.
Remove Iron from the pool water
If your pool is already full and is challenged with iron problems, there are options to physically remove iron from the pool. Most solutions in the pool business are a two-part process. The first part is to sequester the metals–which basically means to cluster metal ions together into larger particle sizes–and then use a filter that can capture the sequestered metals. Such products do exist on the market already.
If you have a D.E. filter, it could be sufficient in and of itself to capture sequestered metals. Removal would be as easy as doing a media replacement. This method has difficulty removing already-oxidized iron, however. Ferric iron, for example, is not as easy to sequester as ferrous iron. More on that in a moment. In order for this plan to work on existing stains, you would need a way of lifting those stains and getting iron into a state where it can be more easily sequestered or chelated.
Chelate or Sequester iron to prevent oxidation
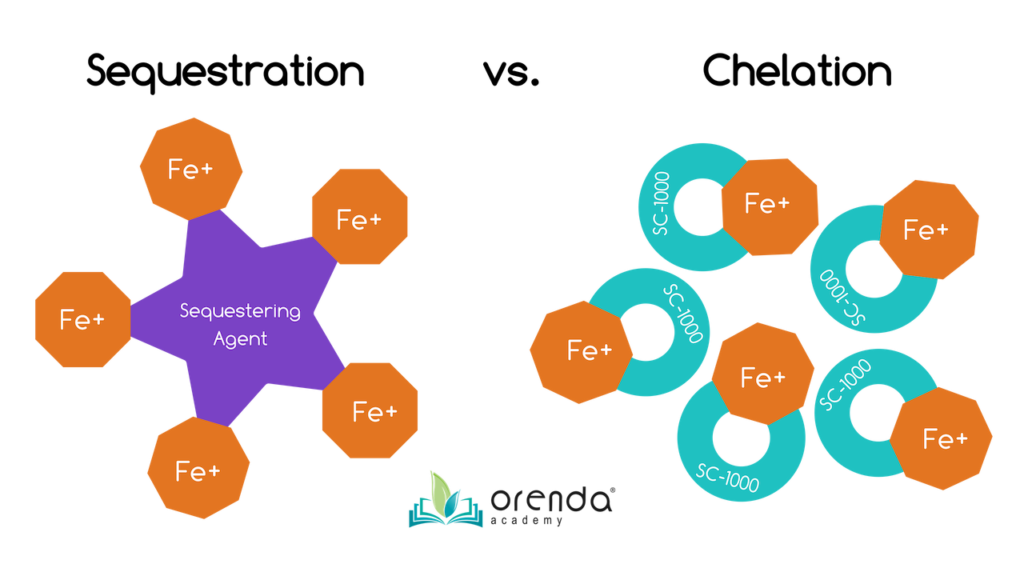
If the problem is not severe enough that it warrants metal removal, you can still prevent oxidation of those metals. Both sequestering agents and chelating agents can bind to metals and hold onto their protons or electrons. If stains already exist, sequestering and chelation is more difficult because the metals are already oxidized and insoluble. An additional step of lifting the stain back into suspension is necessary. To do this, consider using citric acid (also called ascorbic acid, or vitamin C).

Our chelating agent NaturallyFREE is proven to chelate ferric and ferrous iron, but it is very slow to remove stains. It also works better in warmer water. If the water is colder than about 60ºF, the product will be very slow or even dormant. Keep this in mind when using NaturallyFREE.
Sequestering and chelation do not remove metals from the water. They just bind metals to prevent oxidation and staining. You would need some sort of filter to capture and remove the metals. As mentioned before, Regenerative DE filters can capture chelated or sequestered iron. We have even seen it happen on sand filters, but only a few times.
Conclusion
Stains occur when metals get oxidized and become insoluble. Otherwise, metals like iron are in solution or suspension, and mostly invisible. Metals are introduced to our pools through the tap water, with few exceptions like copper algaecide or corroded metal equipment in the pump room. The options to solve this problem are either a strategy of removal, or chemically isolating the metals to prevent further oxidation. In either case, if stains are already present, a citric/ascorbic acid might be needed to lift the stains. Contact us if you need help deciding what to do.


